Categories
- Blog (796)
- Customer Purchase (378)
- Best Sarms stack (6)
- Weight loss peptide (39)
- Other supplements (25)
- Home made (6)
- Testosterone & TRT & AAS (74)
- PCT (27)
When understanding the half-life of a testosterone derivative, we will propose that its half-life is relate to the length of the ester , and the longer the length of the ester chain, the longer the half-life of the corresponding compound. The half-life of testosterone is very short, only a few hours, and the testosterone after the addition of ester has a longer half-life, avoiding the trouble of frequent administration.
The half-life of various testosterone esters
Extending the half-life is of great significance, so does the length of the ester chain only prolong the half-life?
It’s not just that. The length of the attached ester chain also affects the solubility of the compound. Testosterone esters are essentially inject, and increase solubility increases the amount of active compounds contained in the per ml solution.
By testosterone acetate (TAce), testosterone propionate (TPro), testosterone phenylpropionate (TPhp), Experimentation with the solubility of testosterone isocaproate (TIso) has observe that the solubility is correlate with the half-life of the prodrug, so that the acetate with the shortest half-life has the lowest solubility and the isocaproate with the longest half-life has the highest solubility. The longest ester (isocaproate) is about four times more soluble.
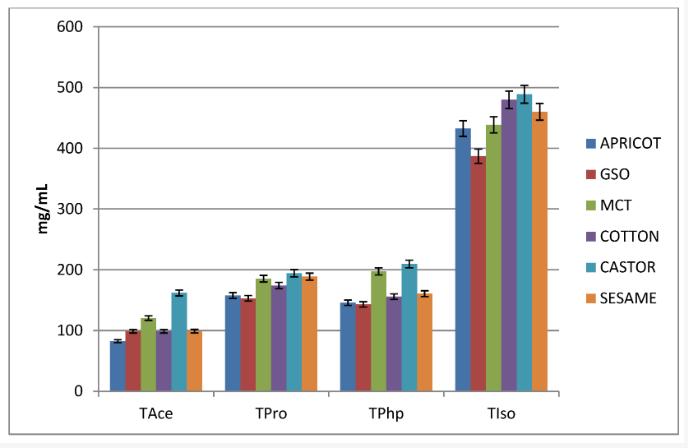
In the experiment, although the phenylpropionate has six more carbon atoms than the propionate, the solubility is similar, so the important factor that increases the solubility is the length of the carbon chain, and the benzene ring does not lead to an increase in solubility.
The longer the ester chain, the higher the solubility of testosterone ester, and the higher the solubility is conducive to improving the content of active ingredients in the finished solution, which is conducive to the stability of the solution, convenient for drug administration, and also to the further preparation of the follow-up.
In experiments, of the six liquids used to dissolve testosterone esters, it was observed that castor oil could support the highest solubility without collapse (compound crystallization) and to a lesser extent MCT (for TAce, TPro, and TPhp), while also acting as a solvent. This is interesting because MCT is characterized by the lowest viscosity coefficient, while castor oil has the highest viscosity.
Therefore, the reasonable selection of testosterone ester and the corresponding solvent can increase the content of active ingredients in the unit dose, and facilitate the user to use testosterone ester better.